
Introduction
Choose the wrong material for a machined implant and the consequences aren't abstract—they show up as stress shielding, implant loosening, or adverse tissue reactions years after surgery. Titanium alloys, PEEK, and 316L stainless steel are the three primary material families used in machined medical implants, and each behaves very differently inside the body.
Material selection shapes implant longevity, biocompatibility, imaging compatibility, and manufacturing cost. Getting it right requires understanding the tradeoffs, not just the properties.
The stakes are substantial. The global implantable medical device market is projected to reach $97.0 billion in 2024, driven largely by orthopedic and spinal surgeries. With that volume comes heightened regulatory and clinical scrutiny—and less tolerance for material decisions made on cost alone. This guide breaks down how titanium, PEEK, and stainless steel compare across biocompatibility, mechanical performance, machinability, and clinical fit.
TLDR
- Titanium alloys (Grade 5 Ti-6Al-4V) deliver superior osseointegration and corrosion resistance for permanent load-bearing implants
- PEEK matches bone elasticity (3–4 GPa) and offers radiolucency, making it the preferred material for spinal fusion cages where imaging clarity matters
- 316L stainless steel provides cost-effectiveness for temporary implants but carries higher corrosion risk and nickel sensitivity concerns
- Permanent load-bearing implants default to titanium; temporary fixation favors 316L stainless steel; spinal and imaging-sensitive applications point to PEEK
Titanium, PEEK, and Stainless Steel: Quick Comparison
Each material brings distinct mechanical and biological trade-offs. The table below maps the properties most critical to implant selection — including stiffness relative to bone, which drives stress shielding risk.
| Property | Titanium (Ti-6Al-4V) | PEEK | 316L Stainless Steel | Human Bone |
|---|---|---|---|---|
| Elastic Modulus | 102-113 GPa | 3-4 GPa | 190-200 GPa | 12-28 GPa (cortical) |
| Density | 4.5 g/cm³ | 1.3 g/cm³ | 8.0 g/cm³ | 1.8-2.0 g/cm³ |
| Corrosion Resistance | Excellent (TiO2 layer) | Excellent (bioinert) | Moderate (pitting risk) | N/A |
| MRI Compatibility | Conditional (artifacts) | Radiolucent (clear imaging) | Conditional (severe artifacts) | N/A |
| Osseointegration | Superior (osteoconductive) | Poor (requires coating) | Limited (fibrous encapsulation) | N/A |
| Relative Cost | High | Very High | Low to Moderate | N/A |
| Common Uses | Hip/knee stems, dental implants | Spinal fusion cages, cranial plates | Temporary fracture fixation | N/A |
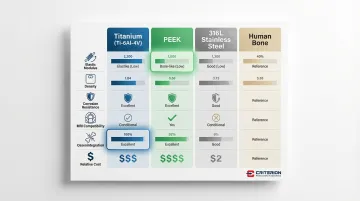
Stainless steel is roughly 10 times stiffer than cortical bone, titanium 4-5 times stiffer. PEEK, at 3-4 GPa, sits closest to bone — a meaningful difference when stress shielding is the primary design concern.
What Are Titanium Alloys and Why Are They the Gold Standard?
What Are Titanium Alloys and Why Do They Dominate Implant Design?
Titanium alloys used in medical implants fall into two primary categories: commercially pure (CP) titanium and Ti-6Al-4V (Grade 5). CP titanium contains minimal impurities and naturally forms a protective titanium dioxide (TiO2) layer when exposed to oxygen. This oxide layer is the key to titanium's exceptional biocompatibility—it's chemically stable, prevents ion release, and actively promotes bone cell attachment.
Grade 5 Ti-6Al-4V contains 6% aluminum and 4% vanadium, delivering nearly twice the tensile strength than CP titanium. The ELI (Extra Low Interstitial) variant limits oxygen, nitrogen, carbon, and hydrogen content, improving ductility and fracture toughness—critical for implants subjected to cyclic loading, like hip stems and spinal hardware.
Three properties explain titanium's dominance in implant design:
- TiO2 surface is osteoconductive, enabling direct bone-to-implant bonding without fibrous tissue formation
- Passive oxide layer stays stable in physiological environments, preventing metal ion release over decades
- Comparable strength to steel at 45% less weight, reducing biomechanical stress on surrounding tissue
These properties apply across both major grades, but the right choice depends on where and how the implant will be loaded.
Grade Comparison
Grade 2 CP Titanium offers better ductility and formability, making it ideal for complex geometries like craniofacial plates and dental implant screws where corrosion resistance matters more than ultimate strength.
Grade 5 Ti-6Al-4V provides higher tensile strength and fatigue resistance, making it the standard for load-bearing implants like hip stems, knee components, and spinal fusion hardware where mechanical demands are extreme.
Use Cases of Titanium Alloys
Titanium dominates permanent implant applications:
- Orthopedic implants - Hip and knee replacements, bone plates, screws, and intramedullary rods
- Dental implants - Root replacements and abutments, with 10-year success rates exceeding 96.4%
- Spinal fusion hardware - Pedicle screws, rods, and interbody cages requiring long-term stability
- Cardiovascular devices - Pacemaker housings and heart valve components
Titanium is preferred for permanent, load-bearing implants where long-term bone integration is critical. The material's bioactive surface actively recruits osteoblasts (bone-forming cells), forming a bond strong enough that retrieval—if ever needed—often requires more force than the surrounding bone can withstand.
What Is PEEK and Why Is It Gaining Traction in Spinal Implants?
PEEK (polyether ether ketone) is a high-performance thermoplastic polymer that received FDA approval for spinal implants in 1998. Unlike metals, it can replace structural components in applications where matching bone's mechanical properties matters more than promoting direct bone bonding.
Core benefits include:
- Elastic modulus of 3-4 GPa closely matches cortical bone (12-28 GPa), reducing stress shielding compared to titanium or stainless steel
- Transparent to X-rays with minimal MRI artifacts, so surgeons can assess bone fusion and surrounding tissue without metal interference
- Highly resistant to chemical degradation in physiological environments, supporting long-term biocompatibility
Important limitations:
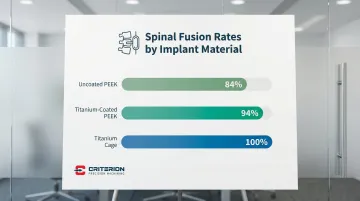
PEEK is bioinert—it doesn't actively bond with bone. In anterior cervical discectomy and fusion (ACDF), titanium cages achieved 100% fusion rates compared to 94% for uncoated PEEK. PEEK also costs more than titanium ($500-$800/kg vs. $18-$40/kg), and its abrasive nature demands specialized machining with precise thermal management to prevent melting or deformation.
Where PEEK Performs Best
PEEK excels when stress shielding and imaging clarity are priorities:
- Spinal fusion cages, where bone-matching stiffness prevents stress shielding and preserves bone density over time
- Cranial reconstruction, where post-operative imaging clarity is essential for tracking healing
- Trauma fixation plates that need enough flexibility to follow bone movement during recovery
Surface treatments address PEEK's osseointegration gap directly. Titanium-coated PEEK achieved 94% fusion rates in transforaminal lumbar interbody fusion (TLIF), compared to 84% for uncoated PEEK. Porous PEEK structures also improve bone attachment by increasing surface area and allowing bone ingrowth.
What Is Stainless Steel and Where Does It Fit in Modern Implants?
316L surgical stainless steel is an iron-chromium-nickel-molybdenum alloy designed for medical applications. The "L" designation indicates low carbon content (reduced from 0.08% to 0.03%), which prevents carbide precipitation during welding and improves corrosion resistance.
Advantages include:
- At $1–$1.50/kg, it's 10–40 times less expensive than titanium or PEEK
- High tensile strength and ductility make it reliable for fracture fixation hardware
- Decades of machining experience and widely available tooling simplify production
- Acceptable biocompatibility for temporary implantation when cost constraints exist
Critical limitations:
- Nickel content (10–14%) is a real concern — nickel hypersensitivity affects 10–15% of the general population and up to 25% of arthroplasty patients
- High elastic modulus (~190 GPa) causes more stress shielding than titanium
- Susceptible to pitting and crevice corrosion in physiological environments
- Gradually releases metal ions over time, unlike titanium's stable oxide layer — a key reason it's avoided in permanent implants
Use Cases of Stainless Steel
316L remains valuable for specific applications:
- Temporary fracture fixation: Plates, screws, and pins removed after bone healing (typically 12–24 months)
- Orthopedic instruments: Surgical tools and guides with less stringent biocompatibility requirements
- Cost-sensitive markets: Applications where budget constraints are the primary driver
For permanent implants, titanium and PEEK have largely displaced 316L — but understanding stainless steel's tradeoffs clarifies exactly why those materials command a higher price and when that premium is justified.
Material Selection: Which Is Best for Your Application?
The right implant material depends on a combination of clinical, anatomical, and economic factors — and the trade-offs look different for every case.
Critical Decision Factors
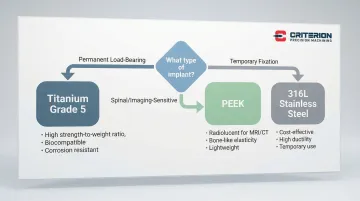
Implant duration and permanence:
- Permanent implants (>2 years): Titanium preferred
- Temporary fixation (<2 years): Stainless steel acceptable
- Long-term but removable: PEEK or titanium depending on imaging needs
Anatomical location and load requirements:
- High load-bearing (hip, knee): Grade 5 titanium
- Moderate load with stress shielding concerns (spine): PEEK
- Low load temporary fixation (hand, foot): 316L stainless steel
Imaging requirements:
- Frequent post-operative imaging needed: PEEK (radiolucent)
- Occasional imaging acceptable: Titanium (manageable artifacts)
- Minimal imaging required: Any material based on other factors
Patient-specific factors:
- Known nickel sensitivity: Avoid 316L
- Poor bone quality: Titanium for better osseointegration
- Revision surgery likely: PEEK for imaging clarity
Budget constraints:
- Premium performance: PEEK or titanium
- Cost-effective temporary solution: 316L stainless steel
- Balanced performance/cost: Grade 2 titanium
Once you've mapped these factors to your case, the recommendations below translate them into material-specific guidance.
Situational Recommendations
Choose Grade 5 Titanium when:
- Maximum strength and osseointegration are required
- Permanent implantation is planned
- Load-bearing demands are high (orthopedic joints, dental implants)
- Long-term clinical success outweighs material cost
Select PEEK when:
- Bone-matching elasticity is critical to prevent stress shielding
- Radiolucent imaging is required for monitoring fusion progress
- Spinal applications where flexibility and imaging clarity matter
- Additive manufacturing is needed for complex, patient-specific geometries
Consider 316L Stainless Steel when:
- Temporary fixation devices will be removed within 12-24 months
- Cost sensitivity is a primary concern
- Patient has no nickel sensitivity history
- Short-term mechanical strength is sufficient for the application
Machining Considerations for Biocompatible Materials
Each material presents unique machining challenges that directly impact implant quality and manufacturing costs.
Material-Specific Challenges
Titanium:
- Low thermal conductivity causes heat buildup at cutting edges
- Work hardening during machining can damage tools and alter surface properties
- Galling (material adhesion to tools) requires specialized cutting geometries
- High-pressure coolant systems essential to manage thermal loads
PEEK:
- Thermoplastic nature makes it sensitive to heat-induced deformation
- Melting point (~343°C) requires precise thermal management
- Abrasive characteristics accelerate tool wear
- Sharp tooling and optimized feed rates prevent dimensional instability
316L Stainless Steel:
- Work hardening during cutting increases tool forces
- Stringy chip formation requires chip-breaking tool geometries
- Proper coolant management prevents thermal damage
- More forgiving than titanium but still demands process control
Tolerance Requirements
Medical implants often require tolerances of ±0.0002" to ±0.001" for critical dimensions. Achieving this precision demands ISO 13485-certified manufacturing processes, advanced CNC equipment, and validated quality control. Not every shop is equipped to meet those standards consistently.

Criterion Precision Machining holds ISO 13485 certification and machines titanium, PEEK, and stainless steel to tolerances down to ±0.0002" using Swiss turning, CNC turning, and 5-axis milling. For "No Failure" implant applications, where microscopic deviations affect clinical outcomes, their ProShop ERP system tracks every operation, tool, and measurement—providing the part-level traceability FDA-registered medical device manufacturing requires.
Conclusion
There is no universal "best" material for medical implants—each offers distinct advantages. Titanium delivers superior osseointegration and corrosion resistance for permanent load-bearing implants. PEEK provides stress-shielding reduction and imaging clarity for spinal applications. Stainless steel offers cost-effective solutions for temporary fixation devices.
The right material choice directly impacts clinical success rates, patient satisfaction, and implant longevity. Each decision involves trade-offs across biomechanical performance, imaging compatibility, regulatory requirements, and cost.
Criterion Precision Machining has machined titanium, PEEK, and stainless steel components for medical device manufacturers since 1953. ISO 13485 certified and holding tolerances down to ±.0002", Criterion works with device engineers from prototype through production — so material selection decisions translate directly into components that perform as designed.
Frequently Asked Questions
What are biocompatible implant materials?
Biocompatible materials function in the human body without triggering toxicity, inflammation, or immune response. The three main categories are titanium alloys, biocompatible polymers like PEEK, and surgical-grade stainless steels—each suited to different load-bearing and longevity requirements.
Which titanium grade is best for implants (Grade 2, 4, or 5)?
Grade 5 Ti-6Al-4V is preferred for high-strength load-bearing applications like hip stems and spinal hardware due to its superior tensile strength and fatigue resistance. Grade 2 CP titanium offers better formability and corrosion resistance for complex geometries and dental applications where ductility matters more than ultimate strength.
Is 316L surgical stainless steel suitable for implants?
316L is suitable for temporary implants and fracture fixation devices intended for removal within 12–24 months. Titanium is preferred for permanent implants—it offers superior corrosion resistance, better osseointegration, and eliminates nickel sensitivity risk, which affects up to 25% of arthroplasty patients.
Which is better for implants, 316L stainless steel or titanium?
Titanium wins for permanent implants: it bonds directly to bone, resists corrosion over decades, and causes less stress shielding than stainless steel. 316L is a practical choice for temporary fixation hardware that will be removed after healing.
What is the most biocompatible material for medical implants?
Titanium and its alloys rank as the most biocompatible metallic materials—their stable TiO2 oxide layer prevents ion release and promotes bone cell attachment. Among polymers, PEEK is the leading option: chemically inert with no metal ion release, though surface modification is needed to support osseointegration.
How does PEEK compare to titanium for spinal implants?
PEEK offers bone-matching elasticity (3-4 GPa vs. 102-113 GPa for titanium), reducing stress shielding and maintaining bone density. Its radiolucency allows clear post-operative imaging without metal artifacts. However, titanium provides superior osseointegration with longer clinical track records—100% fusion rates in some ACDF procedures compared to 94% for uncoated PEEK.


