
Introduction
Surface finish is a critical quality attribute in medical device manufacturing, directly influencing biocompatibility, device longevity, regulatory compliance, and patient safety. A femoral head polished to Ra 0.015 µm will generate minimal wear debris over decades of articulation. The same component finished to Ra 0.05 µm may trigger osteolysis and premature failure.
For surgical instruments, a surface rougher than Ra 0.8 µm becomes a reservoir for biofilm formation, compromising sterilization efficacy and patient outcomes.
Research shows that even nanoscale surface roughness below 30 nm Ra can promote bacterial adhesion, while moderately rough surfaces (Sa 1–2 µm) are essential for osseointegration in bone-contacting implants. This article covers the key standards governing surface finish for implantable devices and surgical instruments, along with practical guidance on specification, measurement, and validation so your components meet both functional and regulatory requirements.
TLDR
- ISO 4287/25178, ASTM F86/F136, and FDA 21 CFR Part 820 govern surface finish for implants and instruments separately
- Ra, Rz, and Sa are the core roughness parameters—each measures a different aspect of surface texture
- Implant finishes span ultra-polished (Ra < 0.05 µm) for articulating surfaces to intentionally rough (Ra 3–6 µm) for osseointegration
- Correct specification depends on functional zone, material, sterilization method, and intended clinical use
Understanding Surface Finish in Medical Device Context
Surface finish refers to the microscopic texture and topography of a component's surface, measured in micrometers (µm) or micro-inches (µin). In medical devices, this goes far beyond aesthetics — it's a functional attribute governing biocompatibility, wear resistance, friction control, corrosion resistance, and cleanability.
Surface finish functions as both a design parameter specified during development and a process output verified during manufacturing. Whether a device passes regulatory review often depends on whether surface characteristics were specified, achieved, and documented correctly.
Functional Impact of Surface Finish on Medical Devices
Surface roughness determines how a medical device interacts with biological tissues and fluids. Four properties drive the most critical outcomes:
- Protein adhesion: Moderately rough surfaces (Sa 1–2 µm) increase expression of FAK (Focal Adhesion Kinase), promoting bone-forming cell response. Ultra-smooth surfaces (Sa < 0.5 µm) minimize protein adhesion — essential for articulating joints where protein buildup accelerates wear.
- Bacterial colonization: Hydrophobic bacteria like S. aureus preferentially colonize rougher surfaces. Surfaces rougher than Ra 0.8 µm retain more organic soil and microbial load even after standard cleaning — a direct infection risk for surgical instruments.
- Tribological performance: Femoral heads require ultra-precision finishes (Ra 0.010–0.015 µm) to minimize polyethylene liner wear. Tolerances outside this range increase wear debris generation, a primary driver of osteolysis and implant failure.
- Sterilization efficacy: Corrosion pits and surface defects harbor biofilms that survive standard sterilization. Electropolishing reduces surface roughness by up to 50% and removes micro-defects, measurably improving reprocessing outcomes for reusable instruments.

Manufacturing and Material Considerations
Different manufacturing processes produce characteristic surface finishes that require active control:
| Process | Typical Ra Range | Medical Application |
|---|---|---|
| CNC Machining | 0.8–3.2 µm | Non-articulating components; requires secondary finishing |
| Mechanical Polishing | < 0.1 µm | Articulating surfaces requiring mirror finishes |
| Electropolishing | Reduces Ra ~50% | Surgical instruments for cleanability and corrosion resistance |
| Additive Manufacturing | 10–30 µm | Too rough for direct use; requires post-processing |
| Plasma Spray (TPS) | ~25 µm | Intentionally rough for mechanical fixation |
Material properties shape what finishing sequences are feasible:
- Titanium alloys — hardness and galling tendency require specialized grinding and polishing sequences before final surface targets are achievable
- Stainless steel 316L — typically reaches Ra 0.4–0.6 µm through mechanical polishing or electropolishing
- Cobalt-chrome — high hardness enables ultra-fine finishes down to Ra ~0.01 µm, making it well-suited for articulating joint surfaces
Key Surface Finish Standards for Medical Devices
Medical device surface finish is governed by an integrated framework of ISO quality management standards, ASTM material specifications, and FDA regulatory requirements. ISO 13485 establishes quality management system requirements, while ASTM F86 addresses surface finish of surgical instruments and ASTM F136 covers titanium implant materials. FDA regulations (21 CFR Part 820) require documented surface finish specifications and verification as part of design controls and manufacturing quality systems.
ISO Standards for Surface Texture Measurement
ISO 4287 and ISO 25178 form the foundation for surface roughness measurement in medical devices. ISO 4287 defines 2D profile parameters measured along a single line:
- Ra (Arithmetic mean deviation): The most common parameter, representing the average absolute deviation from the mean line
- Rz (Maximum height): Measures the vertical distance between the highest peak and lowest valley—critical for detecting protruding asperities that cause abrasive wear
- Rq (Root mean square): More sensitive to outliers than Ra, useful for statistical process control
- RSm (Mean width of elements): Quantifies horizontal spacing of features, useful for textured surfaces
ISO 25178 defines 3D areal parameters that provide more complete characterization for complex implant surfaces:
- Sa (Arithmetic mean height): The 3D equivalent of Ra, essential for specifying osseointegration zones
- Sz (Maximum height): The 3D equivalent of Rz
- Sq (Root mean square height): Used in statistical process control
A single 2D profile can miss critical surface features that only areal measurement captures — particularly for complex geometries like dental implants or porous coatings, where 3D characterization is the more reliable approach.
ISO 1302 provides standardized symbols and conventions for indicating surface finish requirements on technical drawings, so design intent translates without ambiguity into manufacturing specifications.
ASTM Standards for Medical Device Materials
Where ISO standards define how to measure and communicate surface finish, ASTM standards govern the material-level requirements for specific implant alloys.
ASTM F136 covers wrought Ti-6Al-4V ELI (Extra Low Interstitial) alloy for surgical implants. The standard specifies chemical, mechanical, and metallurgical requirements but does not mandate specific numeric Ra values. Mill product can be supplied in various finishes (descaled, pickled, sandblasted, ground, or machined), with final surface finish specifications defined by device manufacturers in engineering drawings.
ASTM F86 is the standard practice for surface preparation and marking of metallic surgical implants. It targets corrosion resistance and biocompatibility by specifying removal of surface contaminants — iron particles, ceramic media, embedded debris — that can act as corrosion initiation sites. The standard also outlines chemical and electrochemical passivation treatments to generate a protective oxide film.
ASTM F2063 addresses wrought Nickel-Titanium (Nitinol) shape memory alloys. The standard allows surfaces to be oxidized, descaled, pickled, blasted, or polished, but explicitly states that mill product is not intended to have the final surface finish of the finished device. Manufacturers must define final polish specifications (often Ra < 0.5 µm) to ensure biocompatibility and fatigue resistance in devices like stents.
Regulatory Documentation Requirements
FDA 21 CFR Part 820 mandates rigorous control over surface finish as a quality attribute:
- Design Controls (820.30): Surface finish requirements must be defined as Design Inputs and verified against Design Outputs
- Process Validation (820.75): Processes like passivation and electropolishing must be validated with high assurance since outcomes cannot be fully verified by non-destructive inspection
- Equipment Calibration (820.72): All inspection equipment must be calibrated against traceable standards

Documentation packages must include specification limits, measurement methods, validation protocols, and acceptance criteria. Manufacturers must establish and justify surface finish specifications based on intended use, risk analysis, and biocompatibility testing. Changes to surface finish specifications may require regulatory notification or approval depending on device classification and approval pathway.
Surface Finish Requirements: Implants vs. Instruments
Medical devices have different functional zones requiring distinct surface finishes based on tissue contact, articulation, and intended lifespan. Each zone carries its own specification logic — getting them wrong affects both performance and regulatory compliance.
Surface Finish for Implantable Devices
Articulating surfaces: Components involved in joint articulation require the highest level of smoothness to prevent wear. Femoral heads typically require Ra 0.010–0.015 µm, while polyethylene acetabular liners feature Ra 0.3–0.5 µm. Hard-on-hard bearings (ceramic or CoCr) often target Ra < 0.025 µm to minimize friction and third-body wear debris.
Osseointegration zones: Surfaces designed for bone anchorage require controlled roughness:
- Smooth abutments: Sa < 0.5 µm to prevent bacterial accumulation at soft tissue interfaces
- Minimally rough: Sa 0.5–1.0 µm
- Moderately rough (optimal): Sa 1.0–2.0 µm, widely cited as optimal for osseointegration in dental and orthopedic implants
- Rough (plasma sprayed): Sa > 2.0 µm; Titanium Plasma Spray coatings achieve Ra ~25 µm for mechanical interlocking
Non-contact surfaces: General implant surfaces not involved in articulation or bone contact typically specify Ra 0.4–1.6 µm for biocompatibility and corrosion resistance.
Surface Finish for Surgical Instruments
Implant zones prioritize biological response. Reusable instruments shift the focus to cleanability and durability across hundreds of sterilization cycles.
Key surface finish requirements for surgical instruments:
- General surfaces: Ra 0.8 µm is the critical cleanability threshold for stainless steel — rougher surfaces retain significantly more organic soil and microbial load. Typical spec: Ra 0.4–0.8 µm.
- Functional surfaces: Cutting edges, gripping surfaces, and articulating joints typically require Ra < 0.2 µm for consistent performance and longevity.
- Sterilization durability: Most manufacturers choose electropolishing over mechanical polishing alone — it reduces surface roughness by roughly 50% while building a passive oxide layer that resists corrosion through repeated autoclave cycles.
Material-Specific Considerations
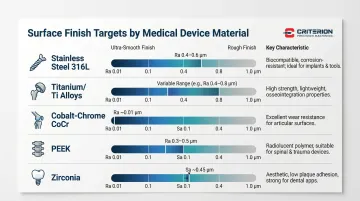
Finish targets vary significantly by material. The table below summarizes achievable Ra values and primary use cases for the most common medical device materials:
| Material | Achievable Ra / Sa | Key Characteristic |
|---|---|---|
| Stainless steel 316L | Ra 0.4–0.6 µm | Electropolishing eliminates micro-defects; preferred for instruments |
| Titanium / Ti alloys | Ra varies; ultra-smooth requires specialized sequences | Hardness and galling tendency demand controlled grinding parameters |
| Cobalt-chrome (CoCr) | Ra ~0.01 µm | High hardness enables ultra-fine finishes; ideal for femoral heads |
| PEEK | Ra 0.3–0.5 µm | Radiolucent; used for spinal cages and liners |
| Zirconia | Sa ~0.45 µm (or intentionally roughened) | Dual-use: smooth for soft tissue, roughened for bone contact in dental implants |
How Surface Finish Is Specified, Measured, and Validated
Surface finish requires three integrated elements: clear specification (what is required), accurate measurement (how to verify), and documented validation (proof of compliance).
Specification Best Practices
Specify surface finish on engineering drawings using ISO 1302 symbols. Include:
- Select Ra, Rz, Sa, or other parameters based on functional requirements
- Define upper and lower limits with appropriate tolerances
- Specify measurement orientation for directional textures
- Define sampling and evaluation lengths explicitly
Critical principle: Specify functional zones separately rather than applying a single finish requirement to an entire component. An orthopedic implant may require Ra < 0.02 µm on articulating surfaces, Ra 3–5 µm on osseointegration zones, and Ra 0.8 µm on non-functional areas.
Including measurement methodology alongside parameter limits prevents inconsistent interpretation across suppliers and inspection teams.
Measurement Methods and Instruments
Contact profilometry is the most common method for 2D surface roughness measurement. A diamond stylus traces the surface while a transducer records vertical displacement.
Key parameters:
- Stylus tip radius: 2 µm recommended for fine finishes (Ra < 0.5 µm); 5 µm for general machining
- Cutoff length (λc): Filter setting separating roughness from waviness; typically 0.25 mm for smooth surfaces, 0.8 mm for machined surfaces
- Evaluation length: Typically five consecutive sampling lengths (e.g., 5 × 0.8 mm = 4.0 mm)
Non-contact optical methods — white light interferometry and confocal microscopy — enable 3D areal surface texture measurement. These are the preferred choice for complex geometries, delicate surfaces, and soft materials like polymers where stylus contact can cause damage.
Regardless of method, vertical resolution must be < 0.01 µm for medical device applications. Calibration standards must be traceable to national standards bodies such as NIST or PTB, with documented calibration frequency. Criterion Precision Machining's ISO 13485-certified quality program uses a Global Advantage CMM with PC-DMIS software to verify surface finish compliance and maintain full measurement traceability.
Validation and Process Control
Initial validation: Demonstrate that manufacturing processes consistently achieve specified surface finishes across the tolerance range. This requires process capability studies showing that the process can reliably produce parts within specification limits.
Statistical process control (SPC): Implement ongoing monitoring of surface finish measurements at defined frequencies. Track trends to detect process drift before it results in out-of-specification parts.
Process capability (Cpk): Studies should demonstrate Cpk > 1.33 for critical surfaces, indicating the manufacturing process is capable of meeting specifications with appropriate margin. For ultra-critical articulating surfaces, Cpk > 1.67 may be required.

Common Specification Errors and How to Avoid Them
Surface finish specifications are often written based on habit or convenience rather than functional requirements, leading to over-specification, measurement confusion, and manufacturing challenges.
Ra Alone Misses Critical Surface Characteristics
Ra is an average value that cannot distinguish between a surface with occasional high peaks (high wear risk) and one with deep valleys (good lubrication retention). Pair Ra with application-specific parameters:
- Articulating surfaces: Add Rz or Rp (maximum peak height) to control abrasive asperities that accelerate wear
- Osseointegration surfaces: Use Sa and Sdr (developed interfacial area ratio) to confirm adequate surface area for bone growth
Ignoring Manufacturing Capability
Specifying Ra < 0.01 µm for a non-articulating handle drives up costs exponentially without adding clinical value. Achieving ultra-smooth finishes requires multiple manual polishing steps or specialized lapping processes, significantly increasing cycle time compared to standard machining.
Omitting Measurement Methodology
Specifying surface finish without defining measurement method, evaluation length, and cutoff wavelength leads to inconsistent measurements and acceptance disputes. Two inspectors using different cutoff lengths can measure significantly different Ra values on the same surface.
Copying Specs from Non-Medical Applications
Surface finish requirements from automotive or aerospace applications may not account for biocompatibility, cleanability, and sterilization requirements critical for medical devices. Always base specifications on medical device standards and functional requirements.
Overlooking Post-Processing Interactions
Surface finish specifications must account for subsequent treatments. Passivation can slightly alter surface texture; sterilization may degrade finishes over repeated cycles. Clarify whether roughness applies pre- or post-treatment, and validate surface characteristics after all processing steps.
Frequently Asked Questions
What does Ra mean for surface finish on medical implants, and how does it differ from Rz?
Ra is the arithmetic average of surface height deviations, providing a general roughness indicator. Rz measures the average maximum peak-to-valley height and is more sensitive to extreme features that may affect wear or failure. Use Ra for general surface quality and Rz when controlling peak height is critical for tribological (wear and friction) performance.
What is the best surface finish and coating for medical implants?
The optimal surface finish depends on implant zone and function. Articulating surfaces require ultra-smooth finishes (Ra < 0.05 µm), while bone-contacting zones need rougher textures (Ra 3–6 µm) for osseointegration. Coatings like titanium plasma spray or hydroxyapatite are then selected based on biocompatibility and integration requirements.
What are the regulatory requirements for surface finish documentation in medical devices?
FDA 21 CFR Part 820 and ISO 13485 require manufacturers to document surface finish specifications, measurement methods, acceptance criteria, and verification records as part of design history files and device master records. All measurement equipment must be calibrated with traceability to national standards.
How do surface finish requirements differ between implantable devices and surgical instruments?
Implants require zone-specific finishes ranging from mirror-polished articulating surfaces (Ra < 0.02 µm) to intentionally roughened osseointegration zones (Ra 3–6 µm), while surgical instruments typically require uniform Ra 0.4–0.8 µm finishes for cleanability and corrosion resistance across all surfaces.
What measurement methods verify surface finish compliance in medical manufacturing?
Contact profilometry (stylus-based) is used for 2D measurements (Ra, Rz), while optical methods (white light interferometry, confocal microscopy) enable 3D areal measurements (Sa, Sz). Selection depends on geometry, material, required resolution, and whether contact measurement might damage the surface.
What surface finish tolerances are acceptable for different medical device applications?
Tolerances vary by application. Orthopedic articulating surfaces typically specify Ra < 0.05 µm with tight tolerances (±0.01 µm). General implant surfaces may specify Ra 0.4–1.6 µm with wider tolerances (±0.2 µm), depending on functional criticality and manufacturing capability.


